Abstract
Leflunomide has recently been approved by the US Food and Drug Administration for the treatment of rheumatoid arthritis. This approval was based on data from double-blind multicentre trials in the US (US 301; leflunomide versus methotrexate versus placebo) and multicentre European trials (leflunomide versus sulfasalazine versus placebo, and leflunomide versus methotrexate versus placebo). In these trials, leflunomide was superior to placebo and similar to methotrexate or sulfasalazine in efficacy and adverse effects. Both methotrexate and leflunomide retarded the rate of radiological progression, entitling them to qualify as disease-modifying agents (DMARDs).
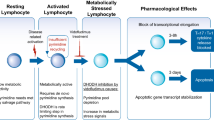
Leflunomide is an immunomodulatory drug that may exert its effects by inhibiting the mitochondrial enzyme dihydro-orotate dehydrogenase (DHO-DH), which plays a key role in the de novo synthesis of the pyrimidine ribonucleotide uridine monophosphate (rUMP). The inhibition of human DHO-DH by A77-1726, the active metabolite of leflunomide, occurs at concentrations (approximately 600 nmol/L) that are achieved during treatment of rheumatoid arthritis. We propose that leflunomide prevents the expansion of activated and autoimmune lymphocytes by interfering with cell cycle progression. This is mediated by inadequate production of rUMP and utilises mechanisms involving the sensor protein p5 3. The relative lack of toxicity of A77 - 1726 on nonlymphoid cells may be due to the ability of these cells to fulfil their ribonucleotide requirements by use of the salvage pyrimidine pathway, which makes them less dependent on de novo synthesis.
Similar content being viewed by others
References
Cao W, Kao P, Chao A, et al. Mechanism of the anti-proliferative action of leflunomide. J Heart Lung Trans 1995; 14: 1016–30
Popovic S, Bartlett R. Disease modifying activity of HWA 486 on the development of SLE in the MRL/lpr mouse. Agents Actions 1986; 19: 313–4
Popovic S, Bartlett R. The use of the murine chronic graft vs host (CGVH) disease, a model for systemic lupus erythematosus (SLE) for drug discovery. Agent Actions 1987; 21: 284–6
Glant T, Milecz K, Bartlett R. Immunomodulation of proteoglycan-induced progressive polyarthritis by leflunomide. Immunopharmacology 1991; 23: 105–16
Stosic-Grujicic S, Dimitrijevic M, Bartlett RR. A novel immunomodulating agent — leflunomide inhibits experimental autoimmune diabetes in mice. Transplant Proc 1996; 28: 3072–3
Bartlett R, Schleyerbach R. Immunopharmacologic properties of a novel isoxazol derivative, HWA 486, with potential antirheumatic activity. I. Disease modifying action on adjuvant arthritis in the rat. Int J Immunopharmacol 1988; 7: 7–18
Thoenes G, Sitter T, Langer K, et al. Leflunomide (HWA 486) inhibits experimental autoimmune tubulointerstitial nephritis in rats. In J Immunopharmacol 1989; 92: 921–9
Ogawa T, Inazu M, Gotoh K, et al. Therapeutic effects of leflunomide, a new antirheumatic drug, on glomerulonephritis induced by anti-basement membrane antibody in rats. Clin Immunol Immunopathol 1991; 6: 103–18
Vidic-Dankovic B, Kosec D, Damjanovic M, et al. Leflunomide prevents the development of experimentally induced myasthenia gravis. Int J Immunopharmacol 1995; 17: 273–81
Bartlett RR, Brendel S, Zielinski T, et al. Leflunomide, an immunorestoring drug for the therapy of autoimmune disorders, especially rheumatoid arthritis. Transplant Proc 1996; 28: 3074–8
Mladenovic V, Domljan Z, Rozman B, et al. Safety and effectiveness of leflunomide in the treatment of patients with active rheumatoid arthritis. Arthritis Rheum 1995; 38: 713–22
Strand V, Cohen S, Schiff M, et al. Treatment of active rheumatoid arthritis with leflunomide compared to placebo and methotrexate. Arch Int Med 1999. In press
Weinblatt M, Kremer J, Coblyn J, et al. Leflunomide plus methotrexate in refractory rheumatoid arthritis: a pilot study [abstract]. Arthritis Rheum 1997; 40: S974
Smolen JS, Kalden JR, Scott DL, et al. Efficacy and safety of leflunomide compared with placebo and sulphasalazine in active rheumatoid arthritis: a double-blind, randomised, multicentre trial. European Leflunomide Study Group [see comments]. Lancet 1999; 353: 259–66
Cherwinski HM, McCarley D, Schatzman R, et al. The immunosuppressant leflunomide inhibits the progression through the cell cycle by a novel mechanism. J Pharm Exp Ther 1995; 272: 460–70
Fairbanks LD, Bofill M, Ruckemann K, et al. Importance of ribonucleotide availability to proliferating T-lymphocytes from healthy humans. Disproportionate expansion of pyrimidine pools and contrasting effects of de novo synthesis inhibitors. J Biol Chem 1995; 270: 29682–9
Silva HT, Cao W, Shorthouse R, et al. Mechanism of action of leflunomide: in vivo uridine administration reverses its inhibition of lymphocyte proliferation. Transplant Proc 1996; 28: 3082–4
Siemasko KF, Chong AS, Williams JW, et al. Regulation of B cell function by the immunosuppressive agent leflunomide. Transplantation 1996; 61: 635–42
Morris R, Huang X, Cao W, et al. Leflunomide (HWA 486) and its analog suppress T and B-cell proliferation in vitro, acute rejection, ongoing rejection and antidonor antibody synthesis in mouse, rat, and cynomolgus monkey recipients. Transplant Proc 1995; 27: 445–7
Linke SP, Clarkin KC, Di Leonardo A, et al. A reversible, p53-dependent G0/G1 cell cycle arrest induced by ribonucleotide depletion in the absence of detectable DNA damage. Genes Dev 1996; 10: 934–47
Herrmann M, Frangou C, Kirschbaum B. Effects of leflunomide on early T-cell signaling and cell cycle commitment. Rheumatol Eur 1997; 26(2): S16
Brazelton TR, Morris RE. Molecular mechanisms of action of new xenobiotic immunosuppressive drugs: tacrolimus (FK506), sirolimus (rapamycin), mycophenolate mofetil and leflunomide. Curr Opin Immunol 1996; 8: 710–20
Weber S, Harnisch L. Use of a population pharmacokinetic model to predict clinical outcome of leflunomide, a new DMARD, in the treatment of rheumatoid arthritis. J Rheumatol 1997; 27:46S
Barankiewicz J, Cohen A. Purine nucleotide metabolism in phytohemagglutinin-induced human T lymphocytes. Arch Biochem Biophys 1987; 258: 167–75
Danska JS, Guidos CJ. Essential and perilous: V(D)J recombination and DNA damage checkpoints in lymphocyte precursors. Semin Immunol 1997; 9: 199–206
Di Leonardo A, Linke SP, Clarkin K, et al. DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Dev 1994; 8: 2540–51
Kubbutat MH, Jones SN, Vousden KH. Regulation of p53 stability by Mdm2. Nature 1997; 387: 299–303
Fontoura B, Sorokina E, David D, et al. p53 is covalently linked to 5.8S rRNA. Mol Cell Biol 1992; 12: 5145–51
Marechall V, Elenbaas B, Piette J, et al. The ribosomal L5 protein is associated with mdm-2 and mdm-2-p53 complexes. Mol Cell Biol 1994; 14: 7414–20
Wahl G, Linke S, Paulson T, et al. Maintaining genetic stability through a p53 mediated checkpoint control. Cancer Survey 1997; 29: 183–219
Lill NL, Grossman SR, Ginsberg D, et al. Binding and modulation of p53 by p300/CBP coactivators. Nature 1997; 387: 823–7
Woo RA, McLure KG, Lees-Miller SP, et al. DNA-dependent protein kinase acts upstream of p53 in response to DNA damage. Nature 1998; 394: 700–4
Lane D. p53, guardian of the genome. Nature 1992; 385: 15–6
Avantaggiati ML, Ogryzko V, Gardner K, et al. Recruitment of p300/CBP in p53-dependent signal pathways. Cell 1997; 89: 1175–84
Levine AJ. p53, the cellular gatekeeper for growth and division. Cell 1997; 88: 323–31
Weinberg R. The retinoblastoma protein and cell cycle control. Cell 1995; 81: 323–30
Wu M, Bellas RE, Shen J, et al. Roles of the tumor suppressor p53 and the cyclin-dependent kinase inhibitor p21WAF1/CIP1 in receptor-mediated apoptosis of WEHI 231 B lymphoma cells. J Exp Med 1998; 187: 1671–9
Deng C, Zhang P, Harper J, et al. Mice lacking p21 undergo normal development, but are defective in G1 checkpoint control. Cell 1995; 82: 675–84
Caelles C, Helmberg A, Karin M. p53 dependent apoptosis in the absence of transcriptional activation of p53 target genes. Nature 1994; 370: 220–3
Green DR, Scott DW. Activation-induced apoptosis in lymphocytes. Curr Opin Immunol 1994; 6: 476–87
Cherwinski HM, Cohn RG, Cheung P, et al. The immunosuppressant leflunomide inhibits lymphocyte proliferation by inhibiting pyrimidine biosynthesis. J Pharmacol Exp Ther 1995; 275: 1043–9
Ruckemann K, Fairbanks L, Carrey E, et al. Leflunomide inhibits pyrimidine de novo synthesis in mitogen stimulated T-lymphocytes from healthy humans. J Biol Chem 1998; 273: 21682–91
Herrmann M, Grangous C, Kirschbaum B. Cell cycle control of the de novo pyrimidine synthesis inhibitor leflunomide through the p53 and p21 pathway. Arthritis Rheum 1997; 40: 123–37
Chatterjee S, Berger N. Growth-phase-dependent response to DNA damage in poly(ADP-ribose) polymerase deficient cell lines: basis for a new hypothesis describing the role of poly(ADP-ribose) polymerase in DNA replication and repair. Mol Cell Biochem 1994; 138: 61–9
Linke SP, Clarkin KC, Wahl GM. p53 mediates permanent arrest over multiple cell cycles in response to gamma-irradiation. Cancer Res 1997; 57: 1171–9
Williamson RA, Yea CM, Robson PA, et al. Dihydroorotate dehydrogenase is a high affinity binding protein for A77 1726 and mediator of a range of biological effects of the immunomodulatory compound. J Biol Chem 1995; 270: 22467–72
Greene S, Watanabe K, Braatz-Trulson J, et al. Inhibition of dihydroorotate dehydrogenase by the immunosuppressive agent leflunomide. Biochem Pharmacol 1995; 50: 861–7
Williamson RA, Yea CM, Robson PA, et al. Dihydroorotate dehydrogenase is a target for the biological effects of leflunomide. Transplant Proc 1996; 28: 3088–91
Kuo EA, Hambleton PT, Kay DP, et al. Synthesis, structure-activity relationships, and pharmacokinetic properties of dihydroorotate dehydrogenase inhibitors: 2-cyano-3-cyclopropyl-3-hydroxy-N-[3′-methyl-4′-(trifluoromethyl)-phenyl] propenamide and related compounds. J Med Chem 1996; 39: 4608–21
Ren S, Wu SK, Lien EJ. Dihydroorotate dehydrogenase inhibitors: quantitative structure-activity relationship analysis. Pharm Res 1998; 15: 286–95
Peters GJ, Schwartsmann G, Nadal JC, et al. In vivo inhibition of the pyrimidine de novo enzyme dihydroorotic acid dehydrogenase by brequinar sodium (DUP-785; NSC 368390) in mice and patients. Cancer Res 1990; 50: 4644–9
Bader B, Knecht W, Fries M, et al. Expression, purification, and characterization of histidine-tagged rat and human flavoenzyme dihydroorotate dehydrogenase. Protein Exp Purif 1998; 13: 414–22
Knecht W, Loffler M. Species related inhibition of human and rat dihydroorotate dehydrogenase by immunosuppressive isoxazol and cinchonic acid derivatives. Biochem Pharmacol 1998. In press
Peters GJ, Laurensse E, de Kant E, et al. The relationship between dihydroorotic acid dehydrogenase and in vitro and in vivo cytostatic effects of brequinar sodium (DUP-785; NSC 368390). Adv Exp Med Biol 1989; 375–82
Silva Jr HT, Cao W, Shorthouse RA, et al. In vitro and in vivo effects of leflunomide, brequinar, and cyclosporine onpyrimidine biosynthesis. Transplant Proc 1997; 29: 1292–3
Huang LC, Clarkin KC, Wahl GM. Sensitivity and selectivity of the DNA damage sensor responsible for activating p53-dependent G1 arrest. Proc Natl Acad Sci USA 1996; 93: 4827–32
Kent EF, Crawford J, Choen HJ, et al. Development of multiple monoclonal serum immunoglobulins (multiclonal gammopathy) following both HLA-identical unfractionated and T cell-depleted haploidentical bone marrow transplantation in severe combined immunodeficiency. J Clin Immunol 1990; 10: 106–13
Cao WW, Kao PN, Aoki Y, et al. A novel mechanism of action of the immunomodulatory drug, leflunomide: augmentation of the immunosuppressive cytokine, TGF-beta 1, and suppression of the immunostimulatory cytokine, IL-2. Transplant Proc 1996; 28: 3079–80
Theofilopoulos AN. The basis of autoimmunity: Part I. Mechanisms of aberrant self-recognition. Immunol Today 1995; 16: 90–8
Morris R. New small molecule immunosuppressants for transplantation: review of essential concepts. J Heart Lung Transplant 1993; 12: S275–S6
Pally C, Smith D, Jaffee B, et al. Side effects of brequinar and brequinar analogues, in combination with cyclosporine in the rat. Toxicology 1998; 15: 207–22
Xu X, Blinder L, Shen J, et al. In vivo mechanism by which leflunomide controls lymphoproliferative and autoimmune disease in MRL/MpJ-lpr/lpr mice. J Immunol 1997; 159: 167–74
Elder RT, Xu X, Williams JW, et al. The immunosuppressive metabolite of leflunomide, A77 1726, affects murine T cells through two biochemical mechanisms. J Immunol 1997; 159: 22–7
Kirsch I, Lista F. Lymphocyte-specific genomic instability. Semin Immunol 1997; 9: 207–15
Knecht W, Bergjohann U, Gonski S, et al. Functional expression of a fragment of human dihydroorotate dehydrogenase by means of the baculovirus expression vector system, and kinetic investigation of the purified recombinant enzyme. Eur J Biochem 1996; 240: 292–301
Dimitrijevic M, Bartlett RR. Leflunomide, a novel immunomodulating drug, inhibits homotypic adhesion of mononuclear cells in rheumatoid arthritis. Transplant Proc 1996; 28: 3086–7
Yeh LS, Gregory CR, Griffey SM, et al. Effects of leflunomide and cyclosporine on myocutaneous allograft survival in the rat. Transplantation 1996; 62: 861–3
Salmi M, Rajala P, Jalkanen S. Homing of mucosal leukocytes to joints. Distinct endothelial ligands in synovium mediate leukocyte-subtype specific adhesion. J Clin Invest 1997; 99: 2165–72
Hamilton L, Vojnovic I, Bakhle Y, et al. The anti-inflammatory drug leflunomide inhibits in vitro and in vivo the activity of COX-2 more potently than the induction of COX-2 or iNOS [abstract]. Proc Br Pharmacol Soc 1996; 18: C51
Schorlemmer HU, Kurrle R. Synergistic activity of malononitrilamides with cyclosporine to control and reverse xenograft rejection. Int J Tissue React 1997; 19: 149–56
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fox, R.I., Herrmann, M.L., Frangou, C.G. et al. How Does Leflunomide Modulate the Immune Response in Rheumatoid Arthritis?. BioDrugs 12, 301–315 (1999). https://doi.org/10.2165/00063030-199912040-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00063030-199912040-00007