Abstract
Objective: The efficacy and tolerability of a new combination inhaler containing both salmeterol 50μg and fluticasone 100μg in a single device was compared with the delivery of the two drugs via two separate inhalers in a multicentre, double-blind, double-dummy study.
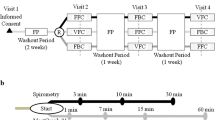
Patients: 244 symptomatic asthma patients (age range 12 to 78 years) were randomised to a 12-week treatment period with either salmeterol/fluticasone (50/100μg twice daily) via a single inhaler (combination) and placebo twice daily via another, or salmeterol 50μg twice daily via one inhaler and fluticasone 100μg twice daily via another (concurrent).
Results: Morning peak expiratory flow rate (PEFR), symptoms and tolerability were collected throughout the treatment period. Adjusted mean improvements in morning PEFR were 42 and 33 L/min for combination and concurrent therapies, respectively, over the 12-week treatment period. Adjusted mean improvements in forced expiratory volume in 1 second (FEV1) from baseline at week 12 were 0.20 and 0.17L for combination and concurrent therapies, respectively. 60% of patients receiving combination inhaler and 64% of those receiving concurrent therapy had a mean daytime symptom score of zero over the treatment period compared with 17 and 15%, respectively, at baseline. Both treatments were well tolerated. Geometric mean morning serum cortisol levels were similar and no differences in the frequency of abnormal results were noted between the two groups.
Conclusion: This was the first study reporting the control of asthma by administration of salmeterol and fluticasone in combination via a single inhaler. The new combination inhaler was as effective and well tolerated as the two drugs administered individually and has potential advantages in terms of convenience.
Similar content being viewed by others
References
British Thoracic Society. The British Guidelines on Asthma Management. Thorax 1997; 52Suppl. 1: S1–S21
Global strategy for asthma management and prevention. WHO/NHLBI Workshop Report. National Institutes of Health, National Heart, Lung and Blood Institute, Publication Number 95–3659, 1995
Chervinsky P, van-As A, Bronsky EA, et al. Fluticasone propionate aerosol for the treatment of adults with mild to moderate asthma. The Fluticasone Propionate Asthma Study Group. J Allergy Clin Immunol 1994; 94: 676–83
Fabbri L, Burge PS, Croonenborgh L, et al. Comparison of fluticasone propionate with beclomethasone dipropionate in moderate to severe asthma treated for one year. International Study Group [see comments]. Thorax 1993; 48: 817–23
Noonan M, Chervinsky P, Busse WW, et al. Fluticasone propionate reduces oral prednisone use while it improves asthma control and quality of life. Am J Respir Crit Care Med 1995; 152: 1467–73
Haahtela T, Jarvinen M, Kava T, et al. Comparison of a beta 2-agonist, terbutaline, with an inhaled corticosteroid, budesonide, in newly detected asthma. N Engl J Med 1991; 325: 388–92
Juniper EF, Kline PA, Vanzieleghem MA, et al. Reduction of budesonide after a year of increased use: a randomized controlled trial to evaluate whether improvements in airway responsiveness and clinical asthma are maintained. J Allergy Clin Immunol 1991; 87: 483–9
Greening AP, Ind PW, Northfield M, et al. Added salmeterol versus higher-dose corticosteroid in asthma patients with symptoms on existing inhaled corticosteroid. Allen & Hanburys Limited UK Study Group. Lancet 1994; 344: 219–24
Woolcock A, Lundback B, Ringdal N, et al. Comparison of addition of salmeterol to inhaled steroids with doubling of the dose of inhaled steroids. Am J Respir Crit Care Med 1996; 153: 1481–8
Ind PW, Dal Negro R, Colman NC, et al. Inhaled fluticasone propionate and salmeterol in moderate adult asthma I: Lung function and symptoms. Am J Respir Crit Care Med 1998; 157: A416
Ind PW, Dal Negro R, Colman NC, et al. Inhaled fluticasone propionate and salmeterol in moderate adult asthma II: Exacerbations. Am J Respir Crit Care Med 1998; 157: A415
Pauwels RA, Löfdahl CG, Postma DS, et al. Effect of inhaled formoterol and budesonide on exacerbations of asthma. N Engl J Med 1997; 337: 1405–11
Boulet LP, Cowie R, Johnston P, et al. Comparison of Diskus inhaler, a new multidose powder inhaler, with Diskhaler inhaler for the delivery of salmeterol to asthmatic patients. Canadian Study Group. J Asthma 1995; 32: 429–36
Rand CS, Wise RA, Nides M, et al. Metered-dose inhaler adherence in a clinical trial. Am Rev Respir Dis 1992; 146: 1559–64
James PN, Anderson JB, Prior JG, et al. Patterns of drug taking in patients with chronic airflow obstruction. Postgrad Med J 1985; 61: 7–10
Kinsman RA, Dirks JF, Dahlem NW. Noncompliance to prescribed-as-needed (PRN) medication use in asthma: usage patterns and patient characteristics. J Psychosom Res 1980; 24: 97–101
Harding JM, Modell M. How patients manage asthma. J R Coll Gen Pract 1985; 35: 226–8
Spector SL, Kinsman R, Mawhinney H, et al. Compliance of patients with asthma with an experimental aerosolized medication: implications for controlled clinical trials. J Allergy Clin Immunol 1986; 77: 65–70
Coutts JAP, Gibson NA, Paton JY. Measuring compliance with inhaled medication in asthma. Arch Dis Child 1992; 67: 332–3
Eisen SA, Miller DK, Woodward RS, et al. The effect of prescribed daily dose frequency on patient medication compliance. Arch Intern Med 1990; 150: 1881–4
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bateman, E.D., Britton, M., Carrillo, J. et al. Salmeterol/Fluticasone Combination Inhaler. Clin. Drug Investig. 16, 193–201 (1998). https://doi.org/10.2165/00044011-199816030-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-199816030-00003