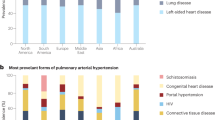
Abstract
Pulmonary arterial hypertension (PAH) is a rare disorder characterized by progressive obliteration of the pulmonary microvasculature that results in elevated pulmonary vascular resistance and premature death. Although no cure exists for PAH, improved understanding of the pathobiological mechanisms of this disease has resulted in the development of effective therapies that target specific aberrant pathways. Agents that modulate abnormalities in the prostacyclin, endothelin, and nitric oxide pathways have been shown in randomized, controlled studies to confer improvements in functional status, pulmonary hemodynamics, and possibly even slow disease progression. Several additional pathways believed to play an important role in the pathogenesis of PAH have been identified as potentially useful therapeutic targets and a number of investigative approaches focusing on these targets are in active development. In this Review, we highlight the pharmacological agents currently available for the treatment of PAH and discuss potential novel strategies.
Key Points
-
Pulmonary arterial hypertension is a complex, rapidly progressive, and incurable disease
-
The past decade has witnessed a remarkable increase in the number of available treatments for pulmonary arterial hypertension that can confer meaningful improvements in important clinical end points
-
Currently licensed therapies for pulmonary arterial hypertension target abnormalities in the endothelin, prostacyclin, and nitric oxide signaling pathways
-
Escalation of therapy using combination regimens is recommended for patients with pulmonary arterial hypertension who continue to exhibit evidence of disease progression
-
Progress in basic and clinical research on pulmonary arterial hypertension has led to improved understanding of disease pathogenesis and identification of a host of novel therapeutic targets
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Humbert, M. et al. Pulmonary arterial hypertension in France: results from a national registry. Am. J. Respir. Crit. Care Med. 173, 1023–1030 (2006).
Peacock, A. J., Murphy, N. F., McMurray, J. J., Caballero, L. & Stewart, S. An epidemiological study of pulmonary arterial hypertension. Eur. Respir. J. 30, 104–109 (2007).
Galiè, N. et al. Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur. Respir. J. 34, 1219–1263 (2009).
Badesch, D. B. et al. Diagnosis and assessment of pulmonary arterial hypertension. J. Am. Coll. Cardiol. 54, S55–S66 (2009).
D'Alonzo, G. E. et al. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann. Intern. Med. 115, 343–349 (1991).
Humbert, M. et al. Survival in patients with idiopathic, familial, and anorexigen-associated pulmonary arterial hypertension in the modern management era. Circulation 122, 156–163 (2010).
Humbert, M., Sitbon, O. & Simonneau, G. Treatment of pulmonary arterial hypertension. N. Engl. J. Med. 351, 1425–1436 (2004).
Galiè, N. et al. A meta-analysis of randomized controlled trials in pulmonary arterial hypertension. Eur. Heart J. 30, 394–403 (2009).
Benza, R. L. et al. Predicting survival in pulmonary arterial hypertension: insights from the Registry to Evaluate Early and Long-Term Pulmonary Arterial Hypertension Disease Management (REVEAL). Circulation 122, 164–172 (2010).
Thenappan, T. et al. Survival in pulmonary arterial hypertension: a reappraisal of the NIH risk stratification equation. Eur. Respir. J. 35, 1079–1087 (2010).
Humbert, M. et al. Survival in incident and prevalent cohorts of patients with pulmonary arterial hypertension. Eur. Respir. J. 36, 549–555 (2010).
Mehta, S. & Shoemaker, G. J. Improving survival in idiopathic pulmonary arterial hypertension: revisiting the “kingdom of the near-dead”. Thorax 60, 981–983 (2005).
Christman, B. W. et al. An imbalance between the excretion of thromboxane and prostacyclin metabolites in pulmonary hypertension. N. Engl. J. Med. 327, 70–75 (1992).
Tuder, R. M. et al. Prostacyclin synthase expression is decreased in lungs from patients with severe pulmonary hypertension. Am. J. Respir. Crit. Care Med. 159, 1925–1932 (1999).
Rubin, L. J. et al. Treatment of primary pulmonary hypertension with continuous intravenous prostacyclin (epoprostenol). Results of a randomized trial. Ann. Intern. Med. 112, 485–491 (1990).
Barst, R. J. et al. Survival in primary pulmonary hypertension with long-term continuous intravenous prostacyclin. Ann. Intern. Med. 121, 409–415 (1994).
Barst, R. J. et al. A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension. The Primary Pulmonary Hypertension Study Group. N. Engl. J. Med. 334, 296–302 (1996).
Shapiro, S. M. et al. Primary pulmonary hypertension: improved long-term effects and survival with continuous intravenous epoprostenol infusion. J. Am. Coll. Cardiol. 30, 343–349 (1997).
McLaughlin, V. V., Shillington, A. & Rich, S. Survival in primary pulmonary hypertension: the impact of epoprostenol therapy. Circulation 106, 1477–1482 (2002).
Simonneau, G. et al. Continuous subcutaneous infusion of treprostinil, a prostacyclin analog, in patients with pulmonary arterial hypertension: a double-blind, randomized, placebo-controlled trial. Am. J. Respir. Crit. Care Med. 165, 800–804 (2002).
Laliberte, K., Arneson, C., Jeffs, R., Hunt, T. & Wade, M. Pharmacokinetics and steady-state bioequivalence of treprostinil sodium (Remodulin) administered by the intravenous and subcutaneous route to normal volunteers. J. Cardiovasc. Pharmacol. 44, 209–214 (2004).
Gomberg-Maitland, M. et al. Transition from intravenous epoprostenol to intravenous treprostinil in pulmonary hypertension. Am. J. Respir. Crit. Care Med. 172, 1586–1589 (2005).
Kallen, A. J. et al. Bloodstream infections in patients given treatment with intravenous prostanoids. Infect. Control Hosp. Epidemiol. 29, 342–349 (2008).
Reisbig, K. A., Coffman, P. A., Floreani, A. A., Bultsma, C. J. & Olsen, K. M. Staggered transition to epoprostenol from treprostinil in pulmonary arterial hypertension. Ann. Pharmacother. 39, 739–743 (2005).
Channick, R. N. et al. Safety and efficacy of inhaled treprostinil as add-on therapy to bosentan in pulmonary arterial hypertension. J. Am. Coll. Cardiol. 48, 1433–1437 (2006).
McLaughlin, V. V. et al. Addition of inhaled treprostinil to oral therapy for pulmonary arterial hypertension: a randomized controlled clinical trial. J. Am. Coll. Cardiol. 55, 1915–22 (2010).
Tapson, V. et al. Results of the FREEDOM-C Study: A Pivotal Study of Oral Treprostinil Used Adjunctively with an ERA and/or PDE5-Inhibitor for the Treatment of PAH [abstract A1040]. Am. J. Respir. Crit. Care Med. 179, A17 (2009).
US NIH. ClinicalTrials.gov. FREEDOM -M: Oral treprostinil as monotherapy for the treatment of pulmonary arterial hypertension (PAH) [online], (2011).
Galiè, N. et al. Effects of beraprost sodium, an oral prostacyclin analog, in patients with pulmonary arterial hypertension: a randomized, double-blind, placebo-controlled trial. J. Am. Coll. Cardiol. 39, 1496–1502 (2002).
Barst, R. J. et al. Beraprost therapy for pulmonary arterial hypertension. J. Am. Coll. Cardiol. 41, 2119–2125 (2003).
Olschewski, H. et al. Inhaled iloprost for severe pulmonary hypertension. N. Engl. J. Med. 347, 322–329 (2002).
Opitz, C. F. et al. Clinical efficacy and survival with first-line inhaled iloprost therapy in patients with idiopathic pulmonary arterial hypertension. Eur. Heart J. 26, 1895–1902 (2005).
Higenbottam, T. W. et al. Treatment of pulmonary hypertension with the continuous infusion of a prostacyclin analog, iloprost. Heart 79, 175–179 (1998).
Higenbottam, T., Butt, A. Y., McMahon, A., Westerbeck, R. & Sharples, L. Long-term intravenous prostaglandin (epoprostenol or iloprost) for treatment of severe pulmonary hypertension. Heart 80, 151–155 (1998).
Olschewski, H. et al. Pharmacodynamics and pharmacokinetics of inhaled iloprost, aerosolized by three different devices, in severe pulmonary hypertension. Chest 124, 1294–1304 (2003).
Opitz, C. F. et al. Assessment of the vasodilator response in primary pulmonary hypertension. Comparing prostacyclin and iloprost administered by either infusion or inhalation. Eur. Heart J. 24, 356–365 (2003).
Hoeper, M. M. et al. Long-term outcome with intravenous iloprost in pulmonary arterial hypertension. Eur. Respir. J. 34, 132–137 (2009).
Giaid, A. et al. Expression of endothelin-1 in the lungs of patients with pulmonary hypertension. N. Engl. J. Med. 328, 1732–1739 (1993).
Seo, B., Oemar, B., Siebenmann, R., von Segesser, L. & Lüscher, T. Both ETA and ETB receptors mediate contraction to endothelin-1 in human blood vessels. Circulation 89, 1203–1208 (1994).
Hirata, Y. et al. Endothelin receptor subtype B mediates synthesis of nitric oxide by cultured bovine endothelial cells. J. Clin. Invest. 91, 1367–1373 (1993).
Channick, R. N. et al. Effects of the dual endothelin-receptor antagonist bosentan in patients with pulmonary hypertension: a randomised placebo-controlled study. Lancet 358, 1119–1123 (2001).
Rubin, L. J. et al. Bosentan therapy for pulmonary arterial hypertension. N. Engl. J. Med. 346, 896–903 (2002).
Humbert, M. et al. Results of European post-marketing surveillance of bosentan in pulmonary hypertension. Eur. Respir. J. 30, 338–344 (2007).
Sitbon, O. et al. Effects of the dual endothelin receptor antagonist bosentan in patients with pulmonary arterial hypertension: a 1-year follow-up study. Chest 124, 247–254 (2003).
Sitbon, O. et al. Survival in patients with class III idiopathic pulmonary arterial hypertension treated with first line oral bosentan compared with an historical cohort of patients started on intravenous epoprostenol. Thorax 60, 1025–1030 (2005).
McLaughlin, V. V. et al. Survival with first-line bosentan in patients with primary pulmonary hypertension. Eur. Respir. J. 25, 244–249 (2005).
Galiè, N. et al. Treatment of patients with mildly symptomatic pulmonary arterial hypertension with bosentan (EARLY study): a double-blind, randomised controlled trial. Lancet 371, 2093–2100 (2008).
Galiè, N. et al. Bosentan therapy in patients with Eisenmenger syndrome: a multicenter, double-blind, randomized, placebo-controlled study. Circulation 114, 48–54 (2006).
Rosenzweig, E. et al. Effects of long-term bosentan in children with pulmonary arterial hypertension. J. Am. Coll. Cardiol. 46, 697–704 (2005).
Sitbon, O. et al. Bosentan for the treatment of human immunodeficiency virus-associated pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 170, 1212–1217 (2004).
Degano, B. et al. Long-term effects of bosentan in patients with HIV-associated pulmonary arterial hypertension. Eur. Respir. J. 33, 92–98 (2009).
Hoeper, M. et al. Bosentan therapy for portopulmonary hypertension. Eur. Respir. J. 25, 502–508 (2005).
Hoeper, M. M. et al. Experience with inhaled iloprost and bosentan in portopulmonary hypertension. Eur. Respir. J. 30, 1096–1102 (2007).
Opitz, C. F., Ewert, R., Kirch, W. & Pittrow, D. Inhibition of endothelin receptors in the treatment of pulmonary arterial hypertension: does selectivity matter? Eur. Heart J. 29, 1936–1948 (2008).
Wu, C. et al. Discovery of TBC11251, a potent, long acting, orally active endothelin receptor-A selective antagonist. J. Med. Chem. 40, 1690–1697 (1997).
Galiè, N. et al. Ambrisentan therapy for pulmonary arterial hypertension. J. Am. Coll. Cardiol. 46, 529–535 (2005).
Galiè, N. et al. Ambrisentan for the treatment of pulmonary arterial hypertension: results of the ambrisentan in pulmonary arterial hypertension, randomized, double-blind, placebo-controlled, multicenter, efficacy (ARIES) study 1 and 2. Circulation 117, 3010–3019 (2008).
McGoon, M. D. et al. Ambrisentan therapy in patients with pulmonary arterial hypertension who discontinued bosentan or sitaxsentan due to liver function test abnormalities. Chest 135, 122–129 (2009).
Oudiz, R. J. et al. Long-term ambrisentan therapy for the treatment of pulmonary arterial hypertension. J. Am. Coll. Cardiol. 54, 1971–1981 (2009).
Barst, R. J. et al. Sitaxsentan therapy for pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 169, 441–447 (2004).
Barst, R. J. et al. Treatment of pulmonary arterial hypertension with the selective endothelin-A receptor antagonist sitaxsentan. J. Am. Coll. Cardiol. 47, 2049–2056 (2006).
Lavelle, A. et al. Sitaxentan-induced hepatic failure in two patients with pulmonary arterial hypertension. Eur. Respir. J. 34, 770–771 (2009).
Lee, W. T. et al. Sitaxentan-related acute liver failure in a patient with pulmonary arterial hypertension. Eur. Respir. J. 37, 472–474 (2011).
Iglarz, M. et al. Pharmacology of macitentan, an orally active tissue-targeting dual endothelin receptor antagonist. J. Pharmacol. Exp. Ther. 327, 736–745 (2008).
US NIH. ClinicalTrials.gov. Study of ACT-064992 on morbidity and mortality in patients with symptomatic pulmonary arterial hypertension [online], (2010).
Giaid, A. & Saleh, D. Reduced expression of endothelial nitric oxide synthase in the lungs of patients with pulmonary hypertension. N. Engl. J. Med. 333, 214–221 (1995).
Galiè, N. et al. Sildenafil citrate therapy for pulmonary arterial hypertension. N. Engl. J. Med. 353, 2148–2157 (2005).
Rubin, L. J. et al. Long-term treatment with sildenafil citrate in pulmonary arterial hypertension: SUPER-2. Chest doi:10.1378/chest.10-0969.
Galiè, N. et al. Tadalafil therapy for pulmonary arterial hypertension. Circulation 119, 2894–2903 (2009).
Barst, R. J. et al. Tadalafil monotherapy and as add-on to background bosentan in patients with pulmonary arterial hypertension. J. Heart Lung Transplant. 30, 632–643 (2011).
O'Callaghan, D. S. & Gaine, S. P. Combination therapy and new types of agents for pulmonary arterial hypertension. Clin. Chest Med. 28, 169–185 (2007).
Hoeper, M. M., Markevych, I., Spiekerkoetter, E., Welte, T. & Niedermeyer, J. Goal-oriented treatment and combination therapy for pulmonary arterial hypertension. Eur. Respir. J. 26, 858–863 (2005).
Galiè, N., Palazzini, M. & Manes, A. Pulmonary arterial hypertension: from the kingdom of the near-dead to multiple clinical trial meta-analyses. Eur. Heart J. 31, 2080–2086 (2010).
US NIH. ClinicalTrials.gov. A study of first-line ambrisentan and tadalafil combination therapy in subjects with pulmonary arterial hypertension (AMBITION) [online], (2011).
Asaki, T., Hamamoto, T., Sugiyama, Y., Kuwano, K. & Kuwabara, K. Structure-activity studies on diphenylpyrazine derivatives: a novel class of prostacyclin receptor agonists. Bioorganic Med. Chem. 15, 6692–6704 (2007).
Kuwano, K., Hashino, A., Noda, K., Kosugi, K. & Kuwabara, K. A long-acting and highly selective prostacyclin receptor agonist prodrug, 2-{4-[(5,6-diphenylpyrazin-2-yl)(isopropyl)amino]butoxy}-N-(methylsulfonyl)acetamide (NS-304), ameliorates rat pulmonary hypertension with unique relaxant responses of its active form, {4-[(5,6-diphenylpyrazin-2-yl)(isopropyl)amino]butoxy}acetic acid (MRE-269), on rat pulmonary artery. J. Pharmacol. Exp. Ther. 326, 691–699 (2008).
Kuwano, K. et al. 2-[4-[(5,6-diphenylpyrazin-2-yl)(isopropyl)amino]butoxy]-N-(methylsulfonyl)acetamide (NS-304), an orally available and long-acting prostacyclin receptor agonist prodrug. J. Pharmacol. Exp. Ther. 322, 1181–1188 (2007).
Simonneau, G. et al. Efficacy, safety and tolerability of ACT-293987, a novel oral, non-prostanoid, prostaglandin I2 (IP) receptor agonist: results from a phase IIa study in pulmonary arterial hypertension (PAH) [abstract A2515]. Am. J. Respir. Crit. Care Med. 181, B16 (2010).
US NIH. ClinicalTrials.gov. ACT-293987 in pulmonary arterial hypertension [online], (2011).
Murthy, K. S., Zhang, K. M., Jin, J. G., Grider, J. R. & Makhlouf, G. M. VIP-mediated G protein-coupled Ca2+ influx activates a constitutive NOS in dispersed gastric muscle cells. Am. J. Physiol. 265, G660–G671 (1993).
Said, S. I. The vasoactive intestinal peptide gene is a key modulator of pulmonary vascular remodeling and inflammation. Ann. N. Y. Acad. Sci. 1144, 148–153 (2008).
Said, S. I. et al. Moderate pulmonary arterial hypertension in male mice lacking the vasoactive intestinal peptide gene. Circulation 115, 1260–1268 (2007).
Petkov, V. et al. Vasoactive intestinal peptide as a new drug for treatment of primary pulmonary hypertension. J. Clin. Invest. 111, 1339–1346 (2003).
Leuchte, H. H. et al. Inhalation of vasoactive intestinal peptide in pulmonary hypertension. Eur. Respir. J. 32, 1289–1294 (2008).
Galiè, N. et al. Effects of inhaled aviptadil (vasoactive intestinal peptide) in patients with pulmonary arterial hypertension (PAH) [abstract A2516]. Am. J. Respir. Crit. Care Med. 181, B16 (2010).
Ghofrani, H. A. & Grimminger, F. Soluble guanylate cyclase stimulation: an emerging option in pulmonary hypertension therapy. Eur. Respir. Rev. 18, 35–41 (2009).
Evgenov, O. V. et al. NO-independent stimulators and activators of soluble guanylate cyclase: discovery and therapeutic potential. Nat. Rev. Drug Discov. 5, 755–768 (2006).
Schermuly, R. T. et al. Expression and function of soluble guanylate cyclase in pulmonary arterial hypertension. Eur. Respir. J. 32, 881–891 (2008).
Ghofrani, H. A. et al. Riociguat for chronic thromboembolic pulmonary hypertension and pulmonary arterial hypertension: a phase II study. Eur. Respir. J. 36, 792–799 (2010).
US NIH. ClinicalTrials.gov. A study to evaluate efficacy and safety of oral BAY63–2521 in patients with pulmonary arterial hypertension (PAH) [online], (2011).
Barst, R. J. PDGF signaling in pulmonary arterial hypertension. J. Clin. Invest. 115, 2691–2694 (2005).
Perros, F. et al. Platelet-derived growth factor expression and function in idiopathic pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 178, 81–88 (2008).
Schermuly, R. T. et al. Reversal of experimental pulmonary hypertension by PDGF inhibition. J. Clin. Invest. 115, 2811–2821 (2005).
Ghofrani, H. A., Seeger, W. & Grimminger, F. Imatinib for the treatment of pulmonary arterial hypertension. N. Engl. J. Med. 353, 1412–1413 (2005).
Patterson, K. C., Weissmann, A., Ahmadi, T. & Farber, H. W. Imatinib mesylate in the treatment of refractory idiopathic pulmonary arterial hypertension. Ann. Intern. Med. 145, 152–153 (2006).
Souza, R., Sitbon, O., Parent, F., Simonneau, G. & Humbert, M. Long-term imatinib treatment in pulmonary arterial hypertension. Thorax 61, 736 (2006).
Ghofrani, H. A. et al. Imatinib in pulmonary arterial hypertension patients with inadequate response to established therapy. Am. J. Respir. Crit. Care Med. 182, 1171–1177 (2010).
US NIH. ClinicalTrials.gov. Imatinib in pulmonary arterial hypertension (IMPRES) [online], (2010).
Moreno-Vinasco, L. et al. Genomic assessment of a multikinase inhibitor, sorafenib, in a rodent model of pulmonary hypertension. Physiol. Genomics 33, 278–291 (2008).
Gomberg-Maitland, M. et al. A dosing/cross-development study of the multikinase inhibitor sorafenib in patients with pulmonary arterial hypertension. Clin. Pharmacol. Ther. 87, 303–310 (2009).
Kerkela, R. et al. Cardiotoxicity of the cancer therapeutic agent imatinib mesylate. Nat. Med. 12, 908–916 (2006).
Chen, M. H., Kerkela, R. & Force, T. Mechanisms of cardiac dysfunction associated with tyrosine kinase inhibitor cancer therapeutics. Circulation 118, 84–95 (2008).
US NIH. ClinicalTrials.gov. Efficacy, safety, tolerability and pharmacokinetics (PK) of nilotinib (AMN107) in pulmonary arterial hypertension [online], (2010).
Abe, K. et al. Tyrosine kinase inhibitors are potent acute pulmonary vasodilators in rats. Am. J. Respir. Cell Mol. Biol. doi:10.1165/rcmb.2010–0371OC.
Dempsie, Y. & MacLean, M. R. Pulmonary hypertension: therapeutic targets within the serotonin system. Br. J. Pharmacol. 155, 455–462 (2008).
Hervé, P. et al. Increased plasma serotonin in primary pulmonary hypertension. Am. J. Med. 99, 249–254 (1995).
Abenhaim, L. et al. Appetite-suppressant drugs and the risk of primary pulmonary hypertension. International Primary Pulmonary Hypertension Study Group. N. Engl. J. Med. 335, 609–616 (1996).
Souza, R. et al. Pulmonary arterial hypertension associated with fenfluramine exposure: report of 109 cases. Eur. Respir. J. 31, 343–348 (2008).
Dumitrascu, R. et al. Terguride ameliorates monocrotaline-induced pulmonary hypertension in rats. Eur. Respir. J. 37, 1104–1118 (2011).
Porvasnik, S. L. et al. PRX-08066, a novel 5-hydroxytryptamine receptor 2B antagonist, reduces monocrotaline-induced pulmonary arterial hypertension and right ventricular hypertrophy in rats. J. Pharmacol. Exp. Ther. 334, 364–372 (2010).
Mouchaers, K. T. et al. Fasudil reduces monocrotaline-induced pulmonary arterial hypertension: comparison with bosentan and sildenafil. Eur. Respir. J. 36, 800–807 (2010).
Fujita, H. et al. Acute vasodilator effects of inhaled fasudil, a specific Rho-kinase inhibitor, in patients with pulmonary arterial hypertension. Heart Vessels 25, 144–149 (2010).
Fukumoto, Y. et al. Acute vasodilator effects of a Rho-kinase inhibitor, fasudil, in patients with severe pulmonary hypertension. Heart 91, 391–392 (2005).
Guilluy, C. et al. RhoA and Rho kinase activation in human pulmonary hypertension: role of 5-HT signaling. Am. J. Resp. Crit. Care Med. 179, 1151–1158 (2009).
Eddahibi, S. et al. Serotonin transporter overexpression is responsible for pulmonary artery smooth muscle hyperplasia in primary pulmonary hypertension. J. Clin. Invest. 108, 1141–1150 (2001).
Kato, T., Hashikabe, H., Iwata, C., Akimoto, K. & Hattori, Y. Statin blocks Rho/Rho-kinase signaling and disrupts the actin cytoskeleton: relationship to enhancement of LPS-mediated nitric oxide synthesis in vascular smooth muscle cells. Biochim. Biophys. Acta 1689, 267–272 (2004).
Nishimura, T. et al. Simvastatin rescues rats from fatal pulmonary hypertension by inducing apoptosis of neointimal smooth muscle cells. Circulation 108, 1640–1645 (2003).
Sun, X. & Ku, D. D. Rosuvastatin provides pleiotropic protection against pulmonary hypertension, right ventricular hypertrophy, and coronary endothelial dysfunction in rats. Am. J. Physiol. Heart Circ. Physiol. 294, H801–H809 (2008).
Wilkins, M. R. et al. Simvastatin as a treatment for pulmonary hypertension trial. Am. J. Respir. Crit. Care Med. 181, 1106–1113 (2010).
Kawut, S. M. et al. Randomized clinical trial of aspirin and simvastatin for pulmonary arterial hypertension: ASA-STAT. Circulation 123, 2985–2993 (2011).
Zhao, Y. D. et al. Rescue of monocrotaline-induced pulmonary arterial hypertension using bone marrow-derived endothelial-like progenitor cells: efficacy of combined cell and eNOS gene therapy in established disease. Circ. Res. 96, 442–450 (2005).
Wang, X. X. et al. Transplantation of autologous endothelial progenitor cells may be beneficial in patients with idiopathic pulmonary arterial hypertension: a pilot randomized controlled trial. J. Am. Coll. Cardiol. 49, 1566–1571 (2007).
US NIH. ClinicalTrials.gov. Pulmonary hypertension: assessment of cell therapy (PHACeT) [online], (2010).
Mereles, D. et al. Exercise and respiratory training improve exercise capacity and quality of life in patients with severe chronic pulmonary hypertension. Circulation 114, 1482–1489 (2006).
Provencher, S. et al. Deleterious effects of beta-blockers on exercise capacity and hemodynamics in patients with portopulmonary hypertension. Gastroenterology 130, 120–126 (2006).
Mohr, L. C. Hypoxia during air travel in adults with pulmonary disease. Am. J. Med. Sci. 335, 71–79 (2008).
Chaouat, A., Weitzenblum, E. & Higenbottam, T. The role of thrombosis in severe pulmonary hypertension. Eur. Respir. J. 9, 356–363 (1996).
Fuster, V. et al. Primary pulmonary hypertension: natural history and the importance of thrombosis. Circulation 70, 580–587 (1984).
Frank, H. et al. The effect of anticoagulant therapy in primary and anorectic drug-induced pulmonary hypertension. Chest 112, 714–721 (1997).
Bonnin, M. et al. Severe pulmonary hypertension during pregnancy: mode of delivery and anesthetic management of 15 consecutive cases. Anesthesiology 102, 1133–1137 (2005).
Sitbon, O. et al. Long-term response to calcium-channel blockers in idiopathic pulmonary arterial hypertension. Circulation 111, 3105–3111 (2005).
Chapelier, A. et al. Comparative outcome of heart-lung and lung transplantation for pulmonary hypertension. J. Thorac. Cardiovasc. Surg. 106, 299–307 (1993).
Kurzyna, M. et al. Atrial septostomy in treatment of end-stage right heart failure in patients with pulmonary hypertension. Chest 131, 977–983 (2007).
Author information
Authors and Affiliations
Contributions
D. S. O'Callaghan, L. Savale, and M. Humbert researched data for the article. D. S. O'Callaghan, L. Savale, D. Montani, X. Jaïs, G. Simonneau, and M. Humbert contributed to the discussion of content. The article was written by D. S. O'Callaghan. All authors reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
L. Savale has received honoraria from Actelion, Novartis, and Pfizer. D. Montani has received honoraria from Actelion, Bayer–Schering, GlaxoSmithKline, Lilly, Novartis, Pfizer, and United Therapeutics and has received research support from Actelion and Novartis. X. Jaïs has received honoraria from Actelion, GlaxoSmithKline, Lilly, and Pfizer. O. Sitbon has acted as a consultant for and has received honoraria and research support from Actelion, GlaxoSmithKline, Lilly, Pfizer, and United Therapeutics. He has also received honoraria and research support from Bayer–Schering and research support from Novartis. Both G. Simonneau and M. Humbert have acted as consultants for and have received honoraria and research support from Actelion, Bayer–Schering, GlaxoSmithKline, Lilly, Novartis, Pfizer, and United Therapeutics. D. S. O'Callaghan declares no competing interests.
Rights and permissions
About this article
Cite this article
O'Callaghan, D., Savale, L., Montani, D. et al. Treatment of pulmonary arterial hypertension with targeted therapies. Nat Rev Cardiol 8, 526–538 (2011). https://doi.org/10.1038/nrcardio.2011.104
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2011.104
This article is cited by
-
The role of RNA m6A methylation in the regulation of postnatal hypoxia-induced pulmonary hypertension
Respiratory Research (2021)
-
Features associated with pulmonary arterial hypertension in Chinese hospitalized systemic lupus erythematosus patients
Clinical Rheumatology (2018)
-
Incidence and risk of respiratory tract infection associated with specific drug therapy in pulmonary arterial hypertension: a systematic review
Scientific Reports (2017)
-
In vitro and in vivo evaluation of dasatinib and imatinib on physiological parameters of pulmonary arterial hypertension
Cancer Chemotherapy and Pharmacology (2017)
-
Plasma Proteomic Study in Pulmonary Arterial Hypertension Associated with Congenital Heart Diseases
Scientific Reports (2016)