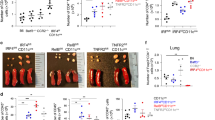
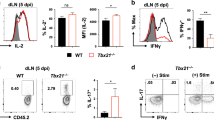
Abstract
The range of regulatory T cell (TR cell) types that control immune responses is poorly understood. We describe here a population of TR cells that developed in vivo from naive CD4+CD25− T cells during a T helper type 1 (TH1)–polarized response, distinct from CD25+ TR cells. These antigen-specific TR cells were induced by CD8α+ DCs, produced both interleukin 10 and interferon-γ, and potently inhibited the development of airway hyper-reactivity. These TR cells expressed the transcription factors Foxp3 and T-bet, indicating that these TR cells are related to TH1 cells. Thus, adaptive TR cells are heterogeneous and comprise TH1-like TR cells as well as previously described TH2-like TR cells, which express Foxp3 and are induced during the development of respiratory tolerance by CD8α− DCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sakaguchi, S., Sakaguchi, N., Asano, M., Itoh, M. & Toda, M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor α-chains (CD25). J. Immunol. 160, 1151–1164 (1995).
Bluestone, J.A. & Abbas, A.K. Natural versus adaptive regulatory T cells. Nat. Rev. Immunol. 3, 253–257 (2003).
Groux, H. et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 389, 737–742 (1997).
Barrat, F.J. et al. In vitro generation of interleukin 10-producing regulatory CD4+ T cells is induced by immunosuppressive drugs and inhibited by T helper type 1 (Th1)- and Th2-inducing cytokines. J. Exp. Med. 195, 603–616 (2002).
Bynoe, M.S., Evans, J.T., Viret, C. & Janeway, C.A. Jr. Epicutaneous immunization with autoantigenic peptides induces T suppressor cells that prevent experimental allergic encephalomyelitis. Immunity 19, 317–328 (2003).
Akbari, O. et al. Antigen-specific regulatory T cells develop via the ICOS-ICOS-ligand pathway and inhibit allergen-induced airway hyperreactivity. Nat. Med. 8, 1024–1032 (2002).
Chen, Y., Kuchroo, V.K., Inobe, J., Hafler, D.A. & Weiner, H.L. Regulatory T cell clones induced by oral tolerance: suppression of autoimmune encephalomyelitis. Science 265, 1237–1240 (1994).
Hansen, G., Yeung, V.P., Berry, G., Umetsu, D.T. & DeKruyff, R.H. Vaccination with heat killed listeria as adjuvant reverses established allergen-induced airway hyperreactivity and inflammation: role of CD8+ T cells and IL-18. J. Immunol. 164, 223–230 (2000).
Hsieh, C.-S. et al. Development of Th1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science 260, 547–549 (1993).
Hsieh, C.S., Macatonia, S.E., O'Garra, A. & Murphy, K.M. Pathogen-induced Th1 phenotype development in CD4+ αβ-TCR transgenic T cells is macrophage dependent. Int. Immunol. 5, 371–382 (1993).
Hansen, G., Berry, G., DeKruyff, R.H. & Umetsu, D.T. Allergen-specific Th1 cells fail to counterbalance Th2 cell-induced airway hyperreactivity but cause severe airway inflammation. J. Clin. Invest. 103, 175–183 (1999).
Randolph, D.A., Carruthers, C.J., Szabo, S.J., Murphy, K.M. & Chaplin, D.D. Modulation of airway inflammation by passive transfer of allergen-specific Th1 and Th2 cells in a mouse model of asthma. J. Immunol. 162, 2375–2383 (1999).
Akbari, O., DeKruyff, R.H. & Umetsu, D.T. Pulmonary dendritic cells secreting IL-10 mediate T cell tolerance induced by respiratory exposure to antigen. Nat. Immunol. 2, 725–731 (2001).
Itoh, M. et al. Thymus and autoimmunity: production of CD25+CD4+ naturally anergic and suppressive T cells as a key function of the thymus in maintaining immunologic self-tolerance. J. Immunol. 162, 5317–5326 (1999).
Lohning, M. et al. Expression of ICOS in vivo defines CD4+ effector T cells with high inflammatory potential and a strong bias for secretion of interleukin 10. J. Exp. Med. 197, 181–193 (2003).
Coyle, A.J. et al. The CD28-related molecule ICOS is required for effective T cell-dependent immune responses. Immunity 13, 95–105 (2000).
Dong, C. et al. ICOS co-stimulatory receptor is essential for T-cell activation and function. Nature 409, 97–101 (2001).
McAdam, A.J. et al. ICOS is critical for CD40-mediated antibody class switching. Nature 409, 102–105 (2001).
Tafuri, A. et al. ICOS is essential for effective T-helper-cell responses. Nature 409, 105–109 (2001).
McAdam, A.J. et al. Mouse inducible costimulatory molecule (ICOS) expression is enhanced by CD28 costimulation and regulates differentiation of CD4+ T cells. J. Immunol. 165, 5035–5040 (2000).
Hori, S., Nomura, T. & Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 299, 1057–1061 (2003).
Yeung, V.P., Gieni, R.S., Umetsu, D.T. & DeKruyff, R.H. Heat killed Listeria monocytogenes as an adjuvant converts established Th2-dominated immune responses into Th1-dominated responses. J. Immunol. 161, 4146–4152 (1998).
Campbell, D., DeKruyff, R.H. & Umetsu, D.T. Allergen immunotherapy: novel approaches in the management of allergic diseases and asthma. Clin. Immunol. 97, 193–202 (2000).
Stock, P. et al. CD8+ T cells regulate immune responses in a murine model of allergen-induced sensitization and airway inflammation. Eur. J. Immunol. 34, 1817–1827 (2004).
Marsland, B.J. et al. Bystander suppression of allergic airway inflammation by lung resident memory CD8+ T cells. Proc. Natl. Acad. Sci. USA 101, 6116–6121 (2004).
Oh, J.W. et al. CD4 T helper cells engineered to produce IL-10 reverse allergen-induced airway hyperreactivity and inflammation. J. Allergy Clin. Immunol. 110, 460–468 (2002).
Hansen, G. et al. CD4+ Th cells engineered To produce latent TGF-β1 reverse allergen-induced airway hyperreactivity and inflammation. J. Clin. Invest. 11, 89–96 (2000).
Gerosa, F. et al. Interleukin-12 primes human CD4 and CD8 T cell clones for high production of both interferon-γ and interleukin-10. J. Exp. Med. 183, 2559–2569 (1996).
Trinchieri, G. Regulatory role of T cells producing both interferon γ and interleukin 10 in persistent infection. J. Exp. Med. 194, F53–57 (2001).
Belkaid, Y. et al. The role of interleukin (IL)-10 in the persistence of Leishmania major in the skin after healing and the therapeutic potential of anti-IL-10 receptor antibody for sterile cure. J. Exp. Med. 194, 1497–1506 (2001).
Pohl-Koppe, A., Balashov, K.E., Steere, A.C., Logigian, E.L. & Hafler, D.A. Identification of a T cell subset capable of both IFN-γ and IL-10 secretion in patients with chronic Borrelia burgdorferi infection. J. Immunol. 160, 1804–1810 (1998).
Gerosa, F. et al. Interleukin-12 primes human CD4 and CD8 T cell clones for high production of both interferon-γ and interleukin-10. J. Exp. Med. 183, 2559–2569 (1996).
Jonuleit, H., Schmitt, E., Schuler, G., Knop, J. & Enk, A.H. Induction of interleukin 10-producing, nonproliferating CD4+ T cells with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J. Exp. Med. 192, 1213–1222 (2000).
Hawiger, D. et al. Dendritic cells induce peripheral T cell unresponsiveness under steady state conditions in vivo. J. Exp. Med. 194, 769–779 (2001).
Steinman, R.M. & Nussenzweig, M.C. Avoiding horror autotoxicus: the importance of dendritic cells in peripheral T cell tolerance. Proc. Natl. Acad. Sci. USA 99, 351–358 (2002).
Bonifaz, L. et al. Efficient targeting of protein antigen to the dendritic cell receptor DEC-205 in the steady state leads to antigen presentation on major histocompatibility complex class I products and peripheral CD8+ T cell tolerance. J. Exp. Med. 196, 1627–1638 (2002).
Inaba, K. et al. Efficient presentation of phagocytosed cellular fragments on the major histocompatibility complex class II products of dendritic cells. J. Exp. Med. 188, 2163–2173 (1998).
Belz, G.T. et al. The CD8α+ dendritic cell is responsible for inducing peripheral self-tolerance to tissue-associated antigens. J. Exp. Med. 196, 1099–1104 (2002).
Iyoda, T. et al. The CD8+ dendritic cell subset selectively endocytoses dying cells in culture and in vivo. J. Exp. Med. 195, 1289–1302 (2002).
Liu, K. et al. Immune tolerance after delivery of dying cells to dendritic cells in situ. J. Exp. Med. 196, 1091–1097 (2002).
Asselin-Paturel, C. et al. Mouse type I IFN-producing cells are immature APCs with plasmacytoid morphology. Nat. Immunol. 2, 1144–1150 (2001).
Wakkach, A. et al. Characterization of dendritic cells that induce tolerance and T regulatory 1 cell differentiation in vivo. Immunity 18, 605–617 (2003).
Krug, A. et al. Interferon-producing cells fail to induce proliferation of naive T cells but can promote expansion and T helper 1 differentiation of antigen-experienced unpolarized T cells. J. Exp. Med. 197, 899–906 (2003).
Martin, P. et al. Characterization of a new subpopulation of mouse CD8α+ B220+ dendritic cells endowed with type 1 interferon production capacity and tolerogenic potential. Blood 100, 383–390 (2002).
McGuirk, P., McCann, C. & Mills, K.H. Pathogen-specific T regulatory 1 cells induced in the respiratory tract by a bacterial molecule that stimulates interleukin 10 production by dendritic cells: a novel strategy for evasion of protective T helper type 1 responses by Bordetella pertussis. J. Exp. Med. 195, 221–231 (2002).
Pulendran, B. et al. Distinct dendritic cell subsets differentially regulate the class of immune response in vivo. Proc. Natl. Acad. Sci. USA 96, 1036–1041 (1999).
Maldonado-Lopez, R. et al. CD8α+ and CD8α− subclasses of dendritic cells direct the development of distinct T helper cells in vivo. J. Exp. Med. 189, 587–592 (1999).
Vieira, P.L. et al. IL-10-secreting regulatory T cells do not express Foxp3 but have comparable regulatory function to naturally occurring CD4+CD25+ regulatory T cells. J. Immunol. 172, 5986–5993 (2004).
Apostolou, I. & Von Boehmer, H. In vivo instruction of suppressor commitment in naive T cells. J. Exp. Med. 199, 1401–1408 (2004).
Bennett, C.L. et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat. Genet. 27, 20–21 (2001).
Wildin, R.S. et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat. Genet. 27, 18–20 (2001).
Chatila, T.A. et al. JM2, encoding a fork head-related protein, is mutated in X-linked autoimmunity-allergic disregulation syndrome. J. Clin. Invest. 106, R75–81 (2000).
Fontenot, J.D., Gavin, M.A. & Rudensky, A.Y. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat. Immunol. 4, 330–336 (2003).
Lyons, A.B. & Parish, C.R. Determination of lymphocyte division by flow cytometry. J. Immunol. Methods 171, 131–137 (1994).
Assenmacher, M., Schmitz, J. & Radbruch, A. Flow cytometric determination of cytokines in activated murine T helper lymphocytes: expression of interleukin-10 in interferon-γ and in interleukin-4-expressing cells. Eur. J. Immunol. 24, 1097–2010 (1994).
Sander, B., Cardell, S. & Möller, E. Interleukin 4 and interferon γ production in restimulated CD4+ and CD8+ cells indicates memory type responsiveness. Scand. J. Immunol. 33, 287–296 (1991).
Szabo, S.J. et al. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell 100, 655–669 (2000).
Hamelmann, E. et al. Noninvasive measurement of airway responsiveness in allergic mice using barometric plethysmography. Am. J. Respir. Crit. Care Med. 156, 766–775 (1997).
Martin, T.R., Gerard, N.P., Galli, S.J. & Drazen, J.M. Pulmonary responses to bronchoconstrictor agonists in the mouse. J. Appl. Physiol. 64, 2318–2323 (1988).
Acknowledgements
This work was supported by National Institutes of Health Public Health Service (AI26322 to D.T.U., HL62348 to D.T.U., HL69507 to R.H.D. and CA84500 and AI39671 to G.J.F.), Deutsche Forschungsgemeinschaft (STO 467/2-1 to P.S.) and American Lung Association of California (O.A.)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
CD8α+ DCs from mice immunized with OVA + HKL show a mature phenotype. (PDF 163 kb)
Supplementary Fig. 2
TH1-like Regulatory cells secrete IL-10. (PDF 490 kb)
Supplementary Table 1
The Number of DO11.10 T Cells Producing IL-10 and IFN-γ Recovered from Recipient Mice (PDF 53 kb)
Rights and permissions
About this article
Cite this article
Stock, P., Akbari, O., Berry, G. et al. Induction of T helper type 1–like regulatory cells that express Foxp3 and protect against airway hyper-reactivity. Nat Immunol 5, 1149–1156 (2004). https://doi.org/10.1038/ni1122
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1122
This article is cited by
-
Integrated single-cell transcriptomics of cerebrospinal fluid cells in treatment-naïve multiple sclerosis
Journal of Neuroinflammation (2022)
-
Single‐cell profiling of lineage determining transcription factors in antigen‐specific CD4+ T cells reveals unexpected complexity in recall responses during immune reconstitution
Immunology & Cell Biology (2017)
-
Induction of eosinophil apoptosis by hydrogen peroxide promotes the resolution of allergic inflammation
Cell Death & Disease (2015)
-
TIM-4, expressed by medullary macrophages, regulates respiratory tolerance by mediating phagocytosis of antigen-specific T cells
Mucosal Immunology (2013)
-
CD8α+β− and CD8α+β+ plasmacytoid dendritic cells induce Foxp3+ regulatory T cells and prevent the induction of airway hyper-reactivity
Mucosal Immunology (2012)