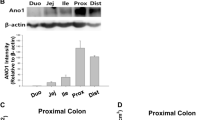
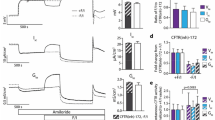
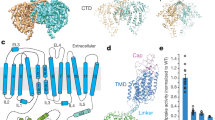
Abstract
It is increasingly being recognized that cells coordinate the activity of separate ion channels that allow electrolytes into the cell. However, a perplexing problem in channel regulation has arisen in the fatal genetic disease cystic fibrosis, which results from the loss of a specific Cl- channel (the CFTR channel) in epithelial cell membranes1. Although this defect clearly inhibits the absorption of Na+ in sweat glands2,3, it is widely accepted that Na+ absorption is abnormally elevated in defective airways in cystic fibrosis4,5. The only frequently cited explanation for this hypertransport is that the activity of an epithelial Na+ channel (ENaC) is inversely related to the activity of the CFTR Cl- channel5,6,7. However, we report here that, in freshly isolated normal sweat ducts, ENaC activity is dependent on, and increases with, CFTR activity. Surprisingly, we also find that the primary defect in Cl- permeability in cystic fibrosis8 is accompanied secondarily by a Na+ conductance in this tissue that cannot be activated. Thus, reduced salt absorption in cystic fibrosis is due not only to poor Cl- conductance but also to poor Na+ conductance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sheppard,D. N. & Welsh,M. J. Structure and function of the CFTR chloride channel. Physiol. Rev. 79, S23–S45 (1999).
Quinton,P. M. Chloride impermeability in cystic fibrosis. Nature 301, 421–422 (1983).
Quinton,P. M. Physiological basis of cystic fibrosis: A historic perspective. Physiol. Rev. 79, S3–S22 (1999).
Boucher,R. C. Human airway ion transport. Part I. Am. J. Respir. Crit. Care Med. 150, 271–281 (1994).
Schwiebert,E. M., Benos,D. J., Egan,M. E., Stutts,M. J. & Guggino,W. B. CFTR is a conductance regulator as well as a chloride channel. Physiol. Rev. 79, S145–S166 (1999).
Stutts,M. J. et al. CFTR as a cAMP-dependent regulator of sodium channels. Science 269, 847–850 (1995).
Kunzelmann,K., Kiser,G. L., Schreiber,R. & Riordan,J. R. Inhibition of epithelial Na+ currents by intracellular domains of the cystic fibrosis transmembrane conductance regulator. FEBS Lett. 400, 341–344 (1997).
Pilewski,J. M. & Frizzell,R. A. Role of CFTR in airway disease. Physiol. Rev. 79, S215–S255 (1999).
Knowles,M. R. et al. Abnormal ion permeation through cystic fibrosis respiratory epithelium. Science 221, 1067–1070 (1983).
Boucher,R. C., Cotton,C. U., Gatzy,J. T., Knowles,M. R. & Yankaskas,J. R. Evidence for reduced Cl- and increased Na+ permeability in cystic fibrosis human primary cell cultures. J. Physiol. (Paris) 405, 77–103 (1988).
Boucher,R. C., Stutts,M. J., Knowles,M. R., Cantley,L. & Gatzy,J. T. Na+ transport in cystic fibrosis respiratory epithelia: abnormal basal rate and response to adenylate cyclase activation. J. Clin. Invest. 78, 1245–1252 (1986).
Stutts,M. J., Rossier,B. C. & Boucher,R. C. Cystic fibrosis transmembrane conductance regulator inverts protein kinase A-mediated regulation of epithelial sodium channel single channel kinetics. J. Biol. Chem. 272, 14037–14040 (1997).
Quinton,P. M. Missing Cl conductance in cystic fibrosis. Am. J. Physiol. 251, C649–C652 (1986).
Quinton,P. M. & Reddy,M. M. Control of CFTR Cl conductance by ATP levels through non-hydrolytic binding. Nature 360, 79–81 (1992).
Reddy,M. M. & Quinton,P. M. Rapid regulation of electrolyte absorption in sweat duct. J. Membr. Biol. 140, 57–67 (1994).
Reddy,M. M. & Quinton,P. M. G-Proteins activate CFTR-GCl in the native sweat duct. Ped. Pulmonol. S14, R46 (1998).
Ismailov,I. I., McDuffie,J. H. & Benos,D. J. Protein kinase A phosphorylation and G-protein regulation of purified renal Na+ channels in planar bilayer membranes. J. Biol. Chem. 269, 10235–10241 (1994).
Flockhart,D. A., Friest,W., Hoppe,J., Loncoln,T. M. & Corban,J. D. ATP analog specificity of cAMP dependent protein kinase, cGMP dependent protein kinase, and phosphorylase kinase. Eur. J. Biochem. 140, 289–295 (1984).
Verrey,F. Antidiuretic hormone action in A6 cells: effect on apical Cl and Na conductances and synergism with aldosterone for NaCl reabsorption. J. Membr. Biol. 138, 65–76 (1994).
Letz,B., Korbmacher,C. cAMP stimulates CFTR-like Cl- channels and inhibits amiloride-sensitive Na+ channels in mouse CCD cells. Am. J. Physiol. 272, C657–C666 (1997).
Ecke,D., Bleich,M. & Greger,R. The amiloride inhibitable Na+ conductance of rat colonic crypt cells is suppressed by forskolin. Pflugers Arch. 431, 984–986 (1996).
Panah,M. R. et al. Hyperexpression of recombinant CFTR in heterologous cells alters its physiological properties. Am. J. Physiol. Cell 274, C310–C318 (1998).
Short,D. B. et al. An apical PDZ protein anchors the cystic fibrosis transmembrane conductance regulator to the cytoskeleton. J. Biol. Chem. 273, 19797–19801 (1998).
Hall,R. A. et al. A C-terminal motif found in the β2-adrenergic receptor, P2Y1 receptor and cystic fibrosis transmembrane conductance regulator determines binding to the Na+/H+ exchanger regulatory factor family of PDZ proteins. Proc. Natl Acad. Sci. USA 95, 8496–8501 (1998).
Jiang,C., Finkbeiner,W. E., Widdicombe,J. H. & Miller,S. S. Fluid transport across cultures of human tracheal glands is altered in cystic fibrosis. J. Physiol. (Lond.) 501, 637–647 (1997).
Welsh,M. J. Electrolyte transport by airway epithelia. Physiol. Rev. 67, 1143–1184 (1987).
Sato,K. & Sato,F. Defective beta adrenergic response of cystic fibrosis sweat glands in vivo and in vitro. J. Clin. Invest. 73, 1763–1771 (1984).
Greger,R. & Schlatter,F. Properties of the basolateral membrane of the cortical thick ascending limb of Henle's loop of rabbit kidney. Eur. J. Physiol. 396, 325–334 (1983).
Acknowledgements
We thank K. Taylor for technical assistance. This work was funded by grants from the Nancy McCracken Endowment, the Cystic Fibrosis Foundation and the USPHS National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reddy, M., Light, M. & Quinton, P. Activation of the epithelial Na+ channel (ENaC) requires CFTR Cl- channel function. Nature 402, 301–304 (1999). https://doi.org/10.1038/46297
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/46297
This article is cited by
-
Downregulation of epithelial sodium channel (ENaC) activity in cystic fibrosis cells by epigenetic targeting
Cellular and Molecular Life Sciences (2022)
-
The Effect of Dynasore Upon the Negative Interaction Between ENaC and CFTR Channels in Xenopus laevis Oocytes
The Journal of Membrane Biology (2022)
-
Measurements of spontaneous CFTR-mediated ion transport without acute channel activation in airway epithelial cultures after modulator exposure
Scientific Reports (2021)
-
ENaC regulation by phospholipids and DGK explained through mathematical modeling
Scientific Reports (2020)
-
Role of CFTR in epithelial physiology
Cellular and Molecular Life Sciences (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.