Abstract
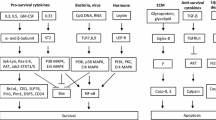
The mainstay of asthma therapy, glucocorticoids (GCs) exert their therapeutic effects through the inhibition of inflammatory signaling and induction of eosinophil apoptosis. However, laboratory and clinical observations of GC-resistant asthma suggest that GCs’ effects on eosinophil viability may depend on the state of eosinophil activation. In the present study we demonstrate that eosinophils stimulated with IL-5 show impaired pro-apoptotic response to GCs. We sought to determine the contribution of GC-mediated transactivating (TA) and transrepressing (TR) pathways in modulation of activated eosinophils’ response to GC by comparing their response to the selective GC receptor (GR) agonist Compound A (CpdA) devoid of TA activity to that upon treatment with Dexamethasone (Dex). IL-5-activated eosinophils showed contrasting responses to CpdA and Dex, as IL-5-treated eosinophils showed no increase in apoptosis compared to cells treated with Dex alone, while CpdA elicited an apoptotic response regardless of IL-5 stimulation. Proteomic analysis revealed that both Nuclear Factor IL-3 (NFIL3) and Map Kinase Phosphatase 1 (MKP1) were inducible by IL-5 and enhanced by Dex; however, CpdA had no effect on NFIL3 and MKP1 expression. We found that inhibiting NFIL3 with specific siRNA or by blocking the IL-5-inducible Pim-1 kinase abrogated the protective effect of IL-5 on Dex-induced apoptosis, indicating crosstalk between IL-5 anti-apoptotic pathways and GR-mediated TA signaling occurring via the NFIL3 molecule. Collectively, these results indicate that (1) GCs’ TA pathway may support eosinophil viability in IL-5-stimulated cells through synergistic upregulation of NFIL3; and (2) functional inhibition of IL-5 signaling (anti-Pim1) or the use of selective GR agonists that don’t upregulate NFIL3 may be effective strategies for the restoring pro-apoptotic effect of GCs on IL-5-activated eosinophils.






Similar content being viewed by others
Abbreviations
- AAD:
-
Aminoactinomycin
- AP1:
-
The activator protein 1
- Bcl2:
-
B-cell lymphoma 2
- BclXL :
-
B-cell lymphoma-extra large
- CpdA:
-
Compound A
- IL:
-
Interleukin
- HES:
-
Hypereosinophilic syndromes
- Jak:
-
Janus tyrosine kinase
- JNK:
-
C-Jun N-terminal kinases
- Mcl-1:
-
Myeloid leukemia cell differentiation protein 1
- MKP1:
-
MAP kinase phosphatase, NFIL3, nuclear factor Interleuki-3 regulated
- Pim1:
-
Proviral integration site for Moloney murine leukemia virus 1
- Stat:
-
Signal transducer and activator of transcription
References
Barnes PJ (1996) Mechanisms of action of glucocorticoids in asthma. Am J Respir Crit Care Med 154(2 Pt 2):S21–S26. doi:10.1164/ajrccm/154.2_Pt_2.S21 discussion S26-27
Klion A (2009) Hypereosinophilic syndrome: current approach to diagnosis and treatment. Annu Rev Med 60:293–306. doi:10.1146/annurev.med.60.062107.090340
Ito K, Chung KF, Adcock IM (2006) Update on glucocorticoid action and resistance. J Allergy Clin Immunol 117(3):522–543. doi:10.1016/j.jaci.2006.01.032
Brode S, Farahi N, Cowburn AS, Juss JK, Condliffe AM, Chilvers ER (2010) Interleukin-5 inhibits glucocorticoid-mediated apoptosis in human eosinophils. Thorax 65(12):1116–1117. doi:10.1136/thx.2009.124909
Hagan JB, Kita H, Gleich GJ (1998) Inhibition of interleukin-5 mediated eosinophil viability by fluticasone 17-propionate: comparison with other glucocorticoids. Clin Exp Allergy 28(8):999–1006
Wallen N, Kita H, Weiler D, Gleich GJ (1991) Glucocorticoids inhibit cytokine-mediated eosinophil survival. J Immunol 147(10):3490–3495
Debierre-Grockiego F, Fuentes V, Prin L, Gouilleux F, Gouilleux-Gruart V (2003) Differential effect of dexamethasone on cell death and STAT5 activation during in vitro eosinopoiesis. Br J Haematol 123(5):933–941
Haldar P, Brightling CE, Hargadon B, Gupta S, Monteiro W, Sousa A, Marshall RP, Bradding P, Green RH, Wardlaw AJ, Pavord ID (2009) Mepolizumab and exacerbations of refractory eosinophilic asthma. N Engl J Med 360(10):973–984. doi:10.1056/NEJMoa0808991
Nair P, Pizzichini MM, Kjarsgaard M, Inman MD, Efthimiadis A, Pizzichini E, Hargreave FE, O’Byrne PM (2009) Mepolizumab for prednisone-dependent asthma with sputum eosinophilia. N Engl J Med 360(10):985–993. doi:10.1056/NEJMoa0805435
Pavord ID, Haldar P, Bradding P, Wardlaw AJ (2010) Mepolizumab in refractory eosinophilic asthma. Thorax 65(4):370. doi:10.1136/thx.2009.122697
Vandevyver S, Dejager L, Tuckermann J, Libert C (2013) New insights into the anti-inflammatory mechanisms of glucocorticoids: an emerging role for glucocorticoid-receptor-mediated transactivation. Endocrinology 154(3):993–1007. doi:10.1210/en.2012-2045
De Bosscher K, Vanden Berghe W, Vermeulen L, Plaisance S, Boone E, Haegeman G (2000) Glucocorticoids repress NF-kappaB-driven genes by disturbing the interaction of p65 with the basal transcription machinery, irrespective of coactivator levels in the cell. Proc Natl Acad Sci USA 97(8):3919–3924
Miller AL, Webb MS, Copik AJ, Wang Y, Johnson BH, Kumar R, Thompson EB (2005) p38 Mitogen-activated protein kinase (MAPK) is a key mediator in glucocorticoid-induced apoptosis of lymphoid cells: correlation between p38 MAPK activation and site-specific phosphorylation of the human glucocorticoid receptor at serine 211. Mol Endocrinol 19(6):1569–1583. doi:10.1210/me.2004-0528
Wu W, Chaudhuri S, Brickley DR, Pang D, Karrison T, Conzen SD (2004) Microarray analysis reveals glucocorticoid-regulated survival genes that are associated with inhibition of apoptosis in breast epithelial cells. Cancer Res 64(5):1757–1764
Robertson S, Allie-Reid F, Vanden Berghe W, Visser K, Binder A, Africander D, Vismer M, De Bosscher K, Hapgood J, Haegeman G, Louw A (2010) Abrogation of glucocorticoid receptor dimerization correlates with dissociated glucocorticoid behavior of compound a. J Biol Chem 285(11):8061–8075. doi:10.1074/jbc.M109.087866
Straub C, Pazdrak K, Young TW, Stafford SJ, Wu Z, Wiktorowicz JE, Haag AM, English RD, Soman KV, Kurosky A (2009) Toward the proteome of the human peripheral blood eosinophil. Proteomics Clin Appl 3(10):1151–1173. doi:10.1002/prca.200900043
Walsh GM, Dewson G, Wardlaw AJ, Levi-Schaffer F, Moqbel R (1998) A comparative study of different methods for the assessment of apoptosis and necrosis in human eosinophils. J Immunol Methods 217(1–2):153–163
Abraham SM, Clark AR (2006) Dual-specificity phosphatase 1: a critical regulator of innate immune responses. Biochem Soc Trans 34(Pt 6):1018–1023. doi:10.1042/BST0341018
Yu YL, Chiang YJ, Yen JJ (2002) GATA factors are essential for transcription of the survival gene E4bp4 and the viability response of interleukin-3 in Ba/F3 hematopoietic cells. J Biol Chem 277(30):27144–27153. doi:10.1074/jbc.M200924200
Kashiwada M, Levy DM, McKeag L, Murray K, Schroder AJ, Canfield SM, Traver G, Rothman PB (2010) IL-4-induced transcription factor NFIL3/E4BP4 controls IgE class switching. Proc Natl Acad Sci USA 107(2):821–826. doi:10.1073/pnas.0909235107
Andina N, Didichenko S, Schmidt-Mende J, Dahinden CA, Simon HU (2009) Proviral integration site for Moloney murine leukemia virus 1, but not phosphatidylinositol-3 kinase, is essential in the antiapoptotic signaling cascade initiated by IL-5 in eosinophils. J Allergy Clin Immunol 123(3):603–611. doi:10.1016/j.jaci.2008.12.004
Stout BA, Bates ME, Liu LY, Farrington NN, Bertics PJ (2004) IL-5 and granulocyte-macrophage colony-stimulating factor activate STAT3 and STAT5 and promote Pim-1 and cyclin D3 protein expression in human eosinophils. J Immunol 173(10):6409–6417
Keeton EK, McEachern K, Dillman KS, Palakurthi S, Cao Y, Grondine MR, Kaur S, Wang S, Chen Y, Wu A, Shen M, Gibbons FD, Lamb ML, Zheng X, Stone RM, Deangelo DJ, Platanias LC, Dakin LA, Chen H, Lyne PD, Huszar D (2014) AZD1208, a potent and selective pan-Pim kinase inhibitor, demonstrates efficacy in preclinical models of acute myeloid leukemia. Blood 123(6):905–913. doi:10.1182/blood-2013-04-495366
Ilmarinen P, Kankaanranta H (2014) Eosinophil apoptosis as a therapeutic target in allergic asthma. Basic Clin Pharmacol Toxicol 114(1):109–117. doi:10.1111/bcpt.12163
Guida L, O’Hehir RE, Hawrylowicz CM (1994) Synergy between dexamethasone and interleukin-5 for the induction of major histocompatibility complex class II expression by human peripheral blood eosinophils. Blood 84(8):2733–2740
Duncan CJ, Lawrie A, Blaylock MG, Douglas JG, Walsh GM (2003) Reduced eosinophil apoptosis in induced sputum correlates with asthma severity. Eur Respir J 22(3):484–490
Gibson PG, Saltos N, Fakes K (2001) Acute anti-inflammatory effects of inhaled budesonide in asthma: a randomized controlled trial. Am J Respir Crit Care Med 163(1):32–36. doi:10.1164/ajrccm.163.1.9807061
Teran LM, Carroll MP, Shute JK, Holgate ST (1999) Interleukin 5 release into asthmatic airways 4 and 24 hours after endobronchial allergen challenge: its relationship with eosinophil recruitment. Cytokine 11(7):518–522. doi:10.1006/cyto.1998.0457
Park SW, Jangm HK, An MH, Min JW, Jang AS, Lee JH, Park CS (2005) Interleukin-13 and interleukin-5 in induced sputum of eosinophilic bronchitis: comparison with asthma. Chest 128(4):1921–1927. doi:10.1378/chest.128.4.1921
Sulakvelidze I, Inman MD, Rerecich T, O’Byrne PM (1998) Increases in airway eosinophils and interleukin-5 with minimal bronchoconstriction during repeated low-dose allergen challenge in atopic asthmatics. Eur Respir J 11(4):821–827
Ueno T, Miyazaki E, Ando M, Nureki S, Kumamoto T (2010) Osteopontin levels are elevated in patients with eosinophilic pneumonia. Respirology 15(7):1111–1121. doi:10.1111/j.1440-1843.2010.01825.x
Kay AB, Klion AD (2004) Anti-interleukin-5 therapy for asthma and hypereosinophilic syndrome. Immunol Allergy Clin North Am 24 (4):645–666, vii. doi:10.1016/j.iac.2004.06.007
Ogbogu PU, Bochner BS, Butterfield JH, Gleich GJ, Huss-Marp J, Kahn JE, Leiferman KM, Nutman TB, Pfab F, Ring J, Rothenberg ME, Roufosse F, Sajous MH, Sheikh J, Simon D, Simon HU, Stein ML, Wardlaw A, Weller PF, Klion AD (2009) Hypereosinophilic syndrome: a multicenter, retrospective analysis of clinical characteristics and response to therapy. J Allergy Clin Immunol 124 (6):1319–1325 e1313. doi:10.1016/j.jaci.2009.09.022
Ikushima S, Inukai T, Inaba T, Nimer SD, Cleveland JL, Look AT (1997) Pivotal role for the NFIL3/E4BP4 transcription factor in interleukin 3-mediated survival of pro-B lymphocytes. Proc Natl Acad Sci USA 94(6):2609–2614
Kirschner AN, Wang J, van der Meer R, Anderson PD, Franco-Coronel OE, Kushner MH, Everett JH, Hameed O, Keeton EK, Ahdesmaki M, Grosskurth SE, Huszar D, Abdulkadir SA (2015) PIM kinase inhibitor AZD1208 for treatment of MYC-driven prostate cancer. J Natl Cancer Inst. doi:10.1093/jnci/dju407
Rothman PB (2010) The transcriptional regulator NFIL3 controls IgE production. Trans Am Clin Climatol Assoc 121:156–171 discussion 171
Mikosz CA, Brickley DR, Sharkey MS, Moran TW, Conzen SD (2001) Glucocorticoid receptor-mediated protection from apoptosis is associated with induction of the serine/threonine survival kinase gene, sgk-1. J Biol Chem 276(20):16649–16654. doi:10.1074/jbc.M010842200
Kristiansen M, Hughes R, Patel P, Jacques TS, Clark AR, Ham J (2010) Mkp1 is a c-Jun target gene that antagonizes JNK-dependent apoptosis in sympathetic neurons. J Neurosci 30(32):10820–10832. doi:10.1523/JNEUROSCI.2824-10.2010
Pazdrak K, Stafford S, Alam R (1995) The activation of the Jak-STAT 1 signaling pathway by IL-5 in eosinophils. J Immunol 155(1):397–402
Stocklin E, Wissler M, Gouilleux F, Groner B (1996) Functional interactions between Stat5 and the glucocorticoid receptor. Nature 383(6602):726–728. doi:10.1038/383726a0
Planey SL, Litwack G (2000) Glucocorticoid-induced apoptosis in lymphocytes. Biochem Biophys Res Commun 279(2):307–312. doi:10.1006/bbrc.2000.3922
Zhang JP, Wong CK, Lam CW (2000) Role of caspases in dexamethasone-induced apoptosis and activation of c-Jun NH2-terminal kinase and p38 mitogen-activated protein kinase in human eosinophils. Clin Exp Immunol 122(1):20–27
Wust S, Tischner D, John M, Tuckermann JP, Menzfeld C, Hanisch UK, van den Brandt J, Luhder F, Reichardt HM (2009) Therapeutic and adverse effects of a non-steroidal glucocorticoid receptor ligand in a mouse model of multiple sclerosis. PLoS ONE 4(12):e8202. doi:10.1371/journal.pone.0008202
De Bosscher K, Vanden Berghe W, Beck IM, Van Molle W, Hennuyer N, Hapgood J, Libert C, Staels B, Louw A, Haegeman G (2005) A fully dissociated compound of plant origin for inflammatory gene repression. Proc Natl Acad Sci USA 102(44):15827–15832. doi:10.1073/pnas.0505554102
De Bosscher K, Vanden Berghe W, Haegeman G (2001) Glucocorticoid repression of AP-1 is not mediated by competition for nuclear coactivators. Mol Endocrinol 15(2):219–227. doi:10.1210/mend.15.2.0591
Yemelyanov A, Czwornog J, Gera L, Joshi S, Chatterton RT Jr, Budunova I (2008) Novel steroid receptor phyto-modulator compound a inhibits growth and survival of prostate cancer cells. Cancer Res 68(12):4763–4773. doi:10.1158/0008-5472.CAN-07-6104
Reber LL, Daubeuf F, Plantinga M, De Cauwer L, Gerlo S, Waelput W, Van Calenbergh S, Tavernier J, Haegeman G, Lambrecht BN, Frossard N, De Bosscher K (2012) A dissociated glucocorticoid receptor modulator reduces airway hyperresponsiveness and inflammation in a mouse model of asthma. J Immunol 188(7):3478–3487. doi:10.4049/jimmunol.1004227
Belvisi MG, Wicks SL, Battram CH, Bottoms SE, Redford JE, Woodman P, Brown TJ, Webber SE, Foster ML (2001) Therapeutic benefit of a dissociated glucocorticoid and the relevance of in vitro separation of transrepression from transactivation activity. J Immunol 166(3):1975–1982
Acknowledgments
We thank Dr. David Konkel (Institute for Translational Sciences, UTMB) for his scientific input and for critical editing of the manuscript. We also thank Dr. Mark Griffin (Microbiology and Immunology, UTMB) for his input on the flow cytometry analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding
This study was supported by the NIH/NCRR KL2RR029875 (to KP) and NIH/NHLBI Proteomics Initiative NO1-HV-28184 (to AK).
Rights and permissions
About this article
Cite this article
Pazdrak, K., Moon, Y., Straub, C. et al. Eosinophil resistance to glucocorticoid-induced apoptosis is mediated by the transcription factor NFIL3. Apoptosis 21, 421–431 (2016). https://doi.org/10.1007/s10495-016-1226-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-016-1226-5